Chemistry
Yenka Chemistry is a complete virtual laboratory. It lets you simulate experiments safely and easily, choosing from over 100 chemicals, and controlling the amounts and concentrations you use.
There’s a wide range of ready-made lessons and activities, and a set of training videos, to help you get started.
It can be used as a flexible demonstration tool – for example, on a whiteboard. Alternatively, students can use Yenka themselves, to learn by experimenting in Yenka’s 3D world.
Inorganic and physical chemistry

Drag chemicals, equipment and glassware from the toolbars at the side of the screen, and combine them as you wish.
Then choose whatever quantities and concentrations you like: reactions are modelled accurately as soon as you mix the chemicals.
The powerful graph tools let you plot data from your reactions – for example, pH against volume. You can also view reaction mechanisms using 3D animations.
The power of simulation
Yenka Inorganic Chemistry is not just a limited set of animations: it’s a full, flexible simulated laboratory.
As well as presenting the models on a whiteboard, you can let your students simulate their own experiments with Yenka Inorganic Chemistry – ideal for coursework, revision or group activities.
Because it’s a virtual lab, they can experiment safely and easily, encouraging scientific enquiry and seeing reactions that aren’t possible or practical in real life.
Electrochemistry simulation
Choose from a range of metal or carbon electrodes, and combine them with one of 28 different electrolytes. For each electrolyte, you can set the concentration and volume precisely.
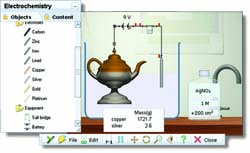
Add a battery and wire it to the electrodes, and watch your simulation run.
Information about the reactions that take place is displayed in the reaction details window. You can look at the reactions and properties at each electrode, as well as at the overall cell equation.
To simulate voltaic cells, use two beakers and a salt bridge, and a voltmeter to measure cell potential.
Make measurements from your simulation using equipment like a thermometer, a pH meter or a balance, or use the graph tool to plot simulated experimental data.
